FDA’s Expanded Access Program
When seriously ill patients face the dilemma of having exhausted all standard of care treatment options, the use of investigational agents may be warranted. However, not all patients qualify for the clinical trials that provide the usual means of gaining access to investigational therapies. In these instances, FDA’s Expanded Access program (also sometimes referred to as “Compassionate Use”), may be allowed for use of investigational drugs, biologics, or medical devices outside of the traditional clinical trial setting for treatment purposes.
However, this is a fairly complex and tightly regulated process whereby physicians and drug companies coordinate the allowable disposition of these treatments for particular patient uses through the Federal Drug Administration (FDA). The AIM team, within Vanderbilt’s Institute of Clinical and Translational Research (VICTR), was created to provide support for navigating the Expanded Access process. For more detailed information on Expanded Access requirements, click on the “Expanded Access Background” tab at the top of this page.Our Goal
Our goal within the VICTR AIM team is to improve efficiency and minimize potential barriers to Expanded Access. We guide the physician, drug company team and other vested parties through the Expanded Access/Compassionate Use process facilitating communication and providing support through all phases, from inquiry to FDA application to delivery of the investigational agent.
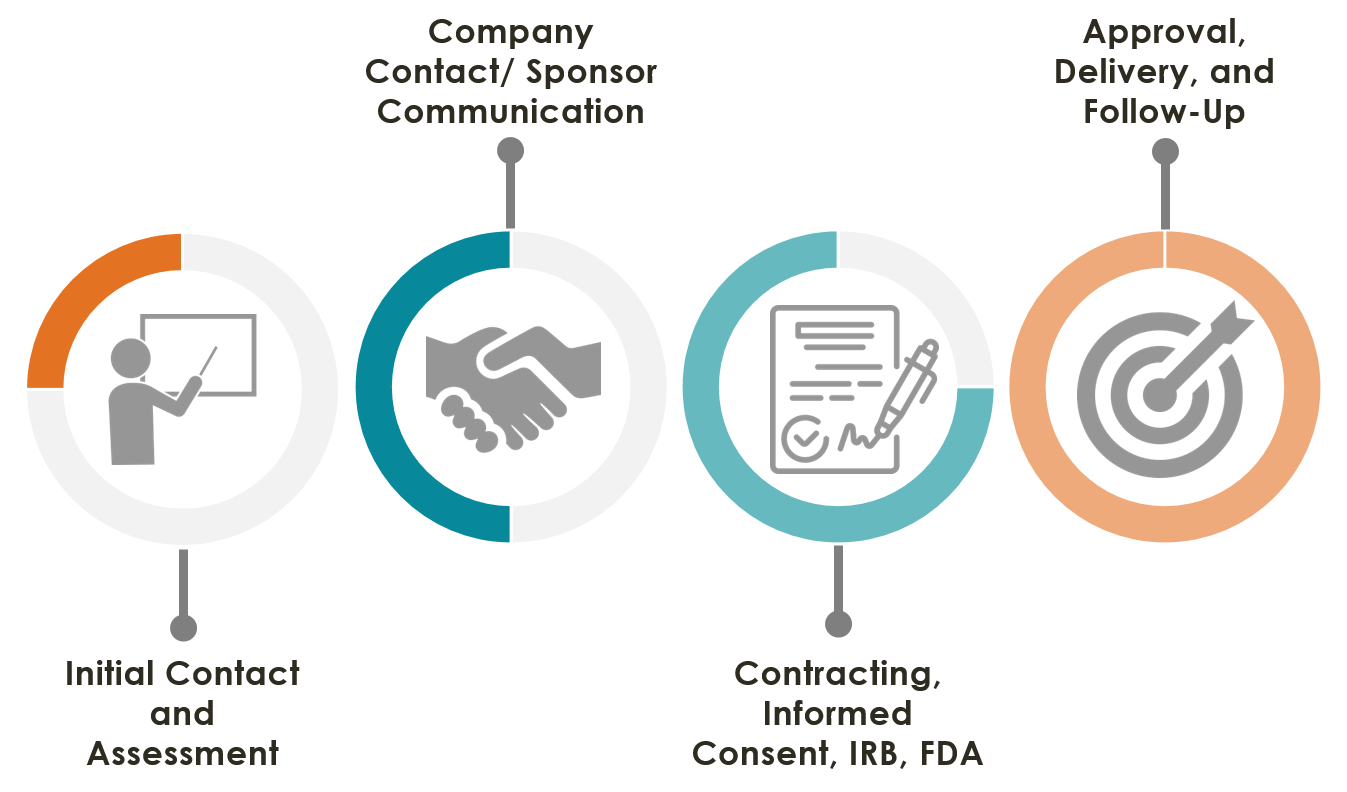
To request AIM support for Expanded Access, click here or click on the AIM Support Request Form link in the navigation bar.
